Note: The descriptions related to development stages are an illustrative image based on planned future development and may differ from the actual progression
Note: 1. AMED: Japan Agency for Medical Research and Development
Note: 2. NMSS: The National Multiple Sclerosis Society
We are currently advancing clinical development of two compounds: nanvuranlat (JPH203) and JPH034.
Nanvuranlat is a novel small-molecule compound identified as a selective inhibitor of LAT1. It competitively binds to the amino acid pocket within LAT1, thereby inhibiting its activity. Importantly, it does not act on LAT2, which is expressed in normal cells, and is known to selectively inhibit LAT1.
Cryo-Electron Microscopy (cryo-EM) structural analysis
Competitive docking of Nanvuranlat(JPH203) to LAT1

Adapted from Y. Lee et al., Nat Commun., 2025, 16, 1635.
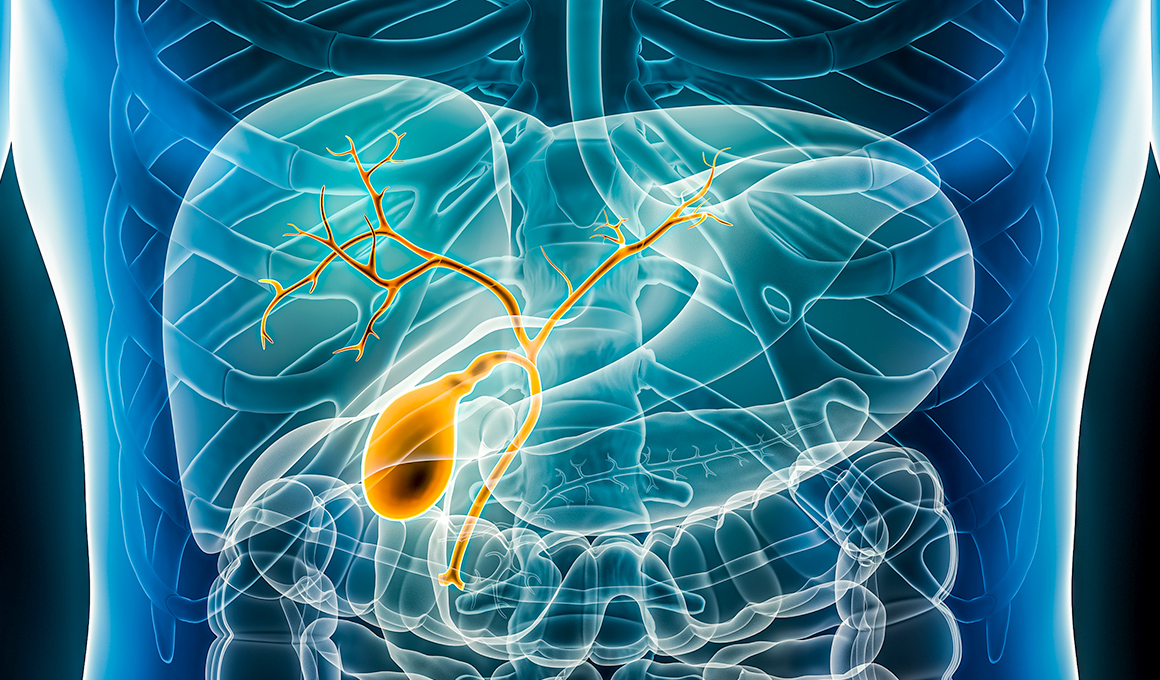
Based on its distinctive tissue distribution, we are advancing the development of nanvuranlat for both second-line and first-line treatment of biliary tract cancer, as well as for colorectal cancer. We have also initiated non-clinical studies in rare diseases to enable future indication expansion.
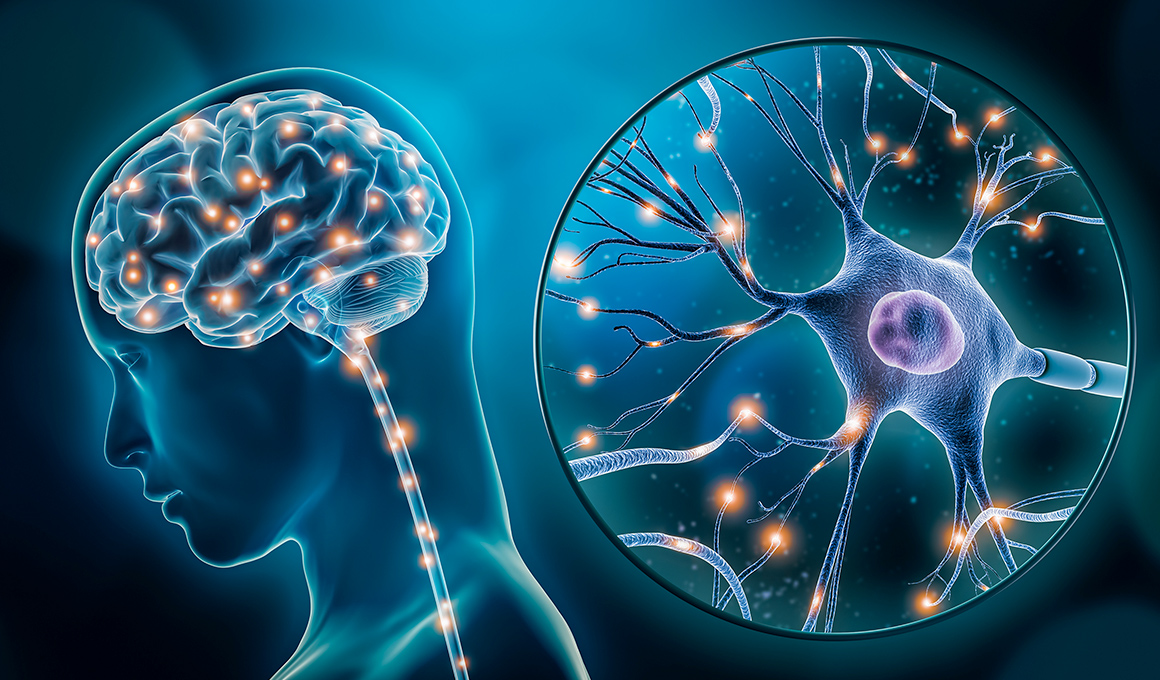
Furthermore, JPH034 is characterized by its high brain penetrance, and we are advancing its development for disorders of the central nervous system, including non-relapsing secondary progressive multiple sclerosis and glioma. In addition, we are engaged in drug discovery research for next-generation LAT1 inhibitors aimed at achieving best-in-class performance and have already identified candidate compounds. Through these efforts, we aim to establish future growth drivers following nanvuranlat and JPH034, thereby achieving sustained business expansion.