Biliary tract cancer(BTC) is diagnosed in approximately 20,000 new patients each year in both Japan and the five major European countries (the United Kingdom, France, Germany, Italy, and Spain), and in about 14,000 patients annually in the United States. Because early-stage disease presents few noticeable symptoms, many patients are currently diagnosed only after the cancer has progressed.
For patients with advanced biliary tract cancer, treatment options remain extremely limited, and the 5-year survival rate is less than 25%, making it one of the cancers with the poorest prognosis after pancreatic cancer. Given this context, biliary tract cancer represents an area of significant unmet medical need, and there is a strong demand for the development of new therapeutic options.
In biliary tract cancer, comparative analyses based on LAT1 expression levels have shown that patients with high LAT1 expression have significantly shorter overall survival.
Comparison of Overall Survival (OS) in BTC (LAT1 High vs. Low Expression)

Kaira et al., BMC Cancer. 2013, 13, 482.
These findings indicate that high LAT1 expression may serve as a prognostic marker in patients with biliary tract cancer and further support LAT1 as a promising candidate for novel molecular targeted therapies.
In Japan, biliary tract cancer is classified into four subtypes: intrahepatic biliary tract cancer, extrahepatic biliary tract cancer, gallbladder cancer, and ampullary cancer. In Western countries, however, ampullary cancer is typically excluded, and the remaining three subtypes are collectively treated as biliary tract cancer.
For first-line treatment, chemotherapy combined with anti–PD-1 or anti–PD-L1 antibodies is used across all subtypes and constitutes the current standard of care. However, fewer than 25% of patients receiving this treatment remain alive at 24 months, indicating a substantial unmet medical need. After approximately 25 weeks of treatment, patients transition to maintenance therapy with either anti–PD-L1 monotherapy or an anti–PD-1 antibody in combination with gemcitabine, and disease progression during this period remains a significant clinical challenge.
In second-line therapy, molecular targeted agents are used, and three such drugs have been approved in the United States. However, only about 30% of patients who progress to second-line treatment carry actionable genetic alterations for which these drugs are effective, such as IDH1 mutations, FGFR2 fusions/positivity, or HER2 positivity. As a result, approximately 70% of patients currently have no effective approved therapies in the United States and Europe, although an approved second-line treatment option is available in Japan.
Treatment options for second-line therapy for biliary tract cancer

*Only those with mutations in each gene (NCCN Guideline BTC 2025, COMPILE Claim Data Dec 2023)
Nanvuranlat has shown combination activity with anti-PD-1/PD-L1 antibodies in animal models. In first-line therapy, adding nanvuranlat to maintenance treatment after week 25 may provide additional therapeutic benefit without competing with the current standard of care.
In second-line therapy, results from previous clinical studies suggest that nanvuranlat has the potential to serve as an effective treatment option for approximately 70% of patients for whom no approved therapies are currently available. Its favorable safety profile further indicates the potential for sustained use, enabling continued support for patients over the course of treatment.
We conducted a Phase II clinical trial of nanvuranlat in Japan. This large-scale, double-blind, randomized, placebo-controlled study screened 211 patients with biliary tract cancer in the second-line or later treatment setting, of whom 105 were randomized. The trial was completed in 2022. Of the randomized patients, 70 were assigned to the nanvuranlat group and 35 to the placebo group. One patient in the nanvuranlat group was subsequently excluded from the analysis due to the identification of an ineligible disease, resulting in 69 patients included in the analysis.
Design of Phase II Clinical Trial in Japan

At the ASCO GI 2023 (American Society of Clinical Oncology Gastrointestinal Cancers Symposium) held in January 2023, the results were presented orally, demonstrating that nanvuranlat achieved the primary endpoint. The drug showed a statistically significant improvement in progression-free survival (PFS) compared with placebo in patients with previously treated, advanced or refractory biliary tract cancer (hazard ratio = 0.56; 95% CI: 0.34–0.90; p = 0.02).
The incidence of treatment-emergent adverse events (AEs) was 41.4% in the nanvuranlat group and 57.1% in the placebo group. Grade 3 or higher AEs occurred in 30.0% of patients in the nanvuranlat group and 22.9% in the placebo group. No events leading to treatment discontinuation, dose reduction, or death were observed in either group, confirming the favorable safety profile of nanvuranlat.Notably, the incidence of Grade 3 or higher AEs was lower than that reported for FOLFOX (69%), which is sometimes used as a second-line therapy for biliary tract cancer, and for the current first-line standard of care—gemcitabine/cisplatin plus durvalumab (76%). These findings highlight a favorable safety profile of nanvuranlat.This characteristic allows for the possibility of long-term treatment and may support future market expansion to patient populations who currently opt for palliative care.
The subgroup analyses from the same study were presented in an oral session at the Clinical Science Symposium of the ASCO Annual Meeting in June 2023. The presentation highlighted that statistically even greater significance was observed in the LAT1 high-expression subgroup (hazard ratio = 0.44; 95% CI: 0.23–0.85; p = 0.01) and in the subgroup of patients with extrahepatic biliary tract cancer or gallbladder cancer (hazard ratio = 0.22; 95% CI: 0.10–0.49; p < 0.001).
Because the acceptance rate for oral presentations at ASCO is only a few percent, the selection of nanvuranlat clinical trial results for two oral presentations in 2023 indicates the high international recognition of our data. The study results were also published in the September 15, 2023 issue of Clinical Cancer Research, a journal of the American Association for Cancer Research (AACR).
Most recently, subgroup analysis results for overall survival (OS) were presented in a poster session at the ESMO Congress 2025. In the subgroup analysis presented, a hazard ratio of 0.76 (95% CI: 0.40–1.26) was observed when focusing on the three biliary tract cancer subtypes excluding ampullary cancer, which is not classified as biliary tract cancer in Western countries.Additionally, in an analysis limited to patients receiving second-line therapy (excluding those treated in third-line and later settings), the hazard ratio was 0.55 (95% CI: 0.09–3.54). Among patients who had not undergone surgery, the hazard ratio was 0.53 (95% CI: 0.28–1.01), demonstrating favorable trends toward improved OS.
Nanvuranlat Phase II Clinical Trial: Subgroup Analysis of OS Hazard Ratios

1. IHC: lntrahepatic biliary tract cancer, EHC: Extra hepatic biliary tract cancer, GBC: Gallbladder cancer
2. The 95% confidence interval represents the range within which the true value is estimated to lie with 95% probability
Furthermore, the poster presentation also introduced an exposure–response analysis conducted using data from the same study. This analysis evaluated the relationship between systemic drug exposure and clinical efficacy. The results demonstrated a positive correlation between accumulated exposure (accumulated AUC) and overall survival (OS). Higher accumulated exposure was also associated with a greater tendency toward tumor shrinkage.
Exposure-Response Analysis of Nanvuranlat
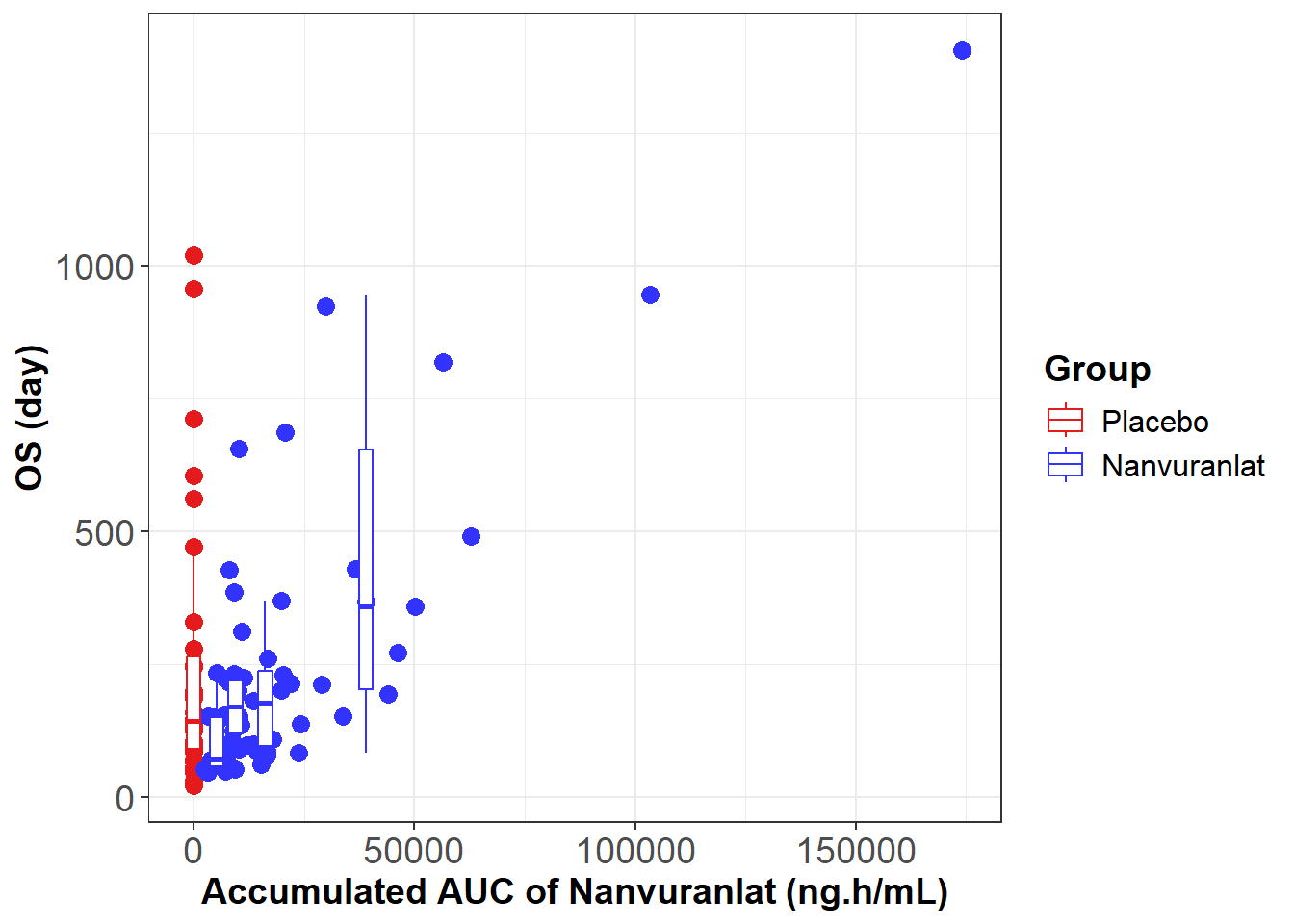
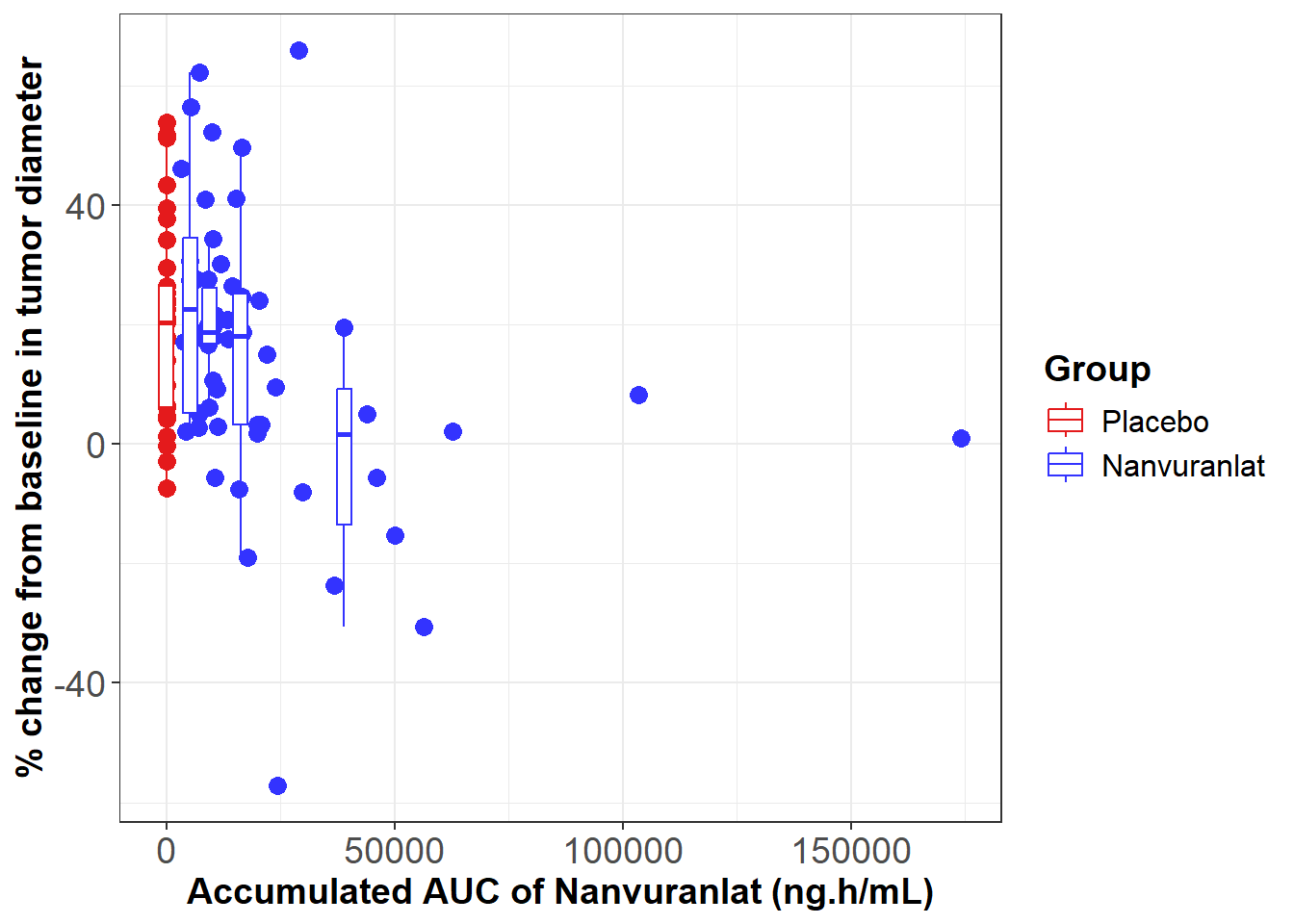
M. Ueno, et al., ESMO 2025. 98P - Subgroup analysis of a Ph.II study of nanvuranlat in pre-treated, advanced, refractory biliary tract cancer patients (BTC) support an enriched patient population for a planned Ph.III study.


Investigator-Initiated Clinical Trial
