
Our corporate philosophy is to "By pursuing new possibilities for SLC transporters and developing innovative new drugs that address unmet medical needs, we will contribute to supporting people around the world to maintain good health and hope." Based on this philosophy, we will work to create innovative pharmaceuticals to achieve sustainable corporate growth and increase social value.
In addition, we have established the following three codes of conduct to realize our corporate philosophy
- Passion to challenge as a global venture company
- The pursuit of science
- Strict adherence to compliance
We have established the following “Goals for 2030” as part of our long-term management strategy and are steadily implementing phased initiatives toward their achievement.
- Obtain global approval for LAT1 inhibitor (Nanvuranlat, First-in-Class) as a monotherapy for BTC
・Aiming for a blockbuster product from Japan (annual sales of USD 1B) - Pursue potential of immune mechanisms, expanding indications into other cancers, autoimmune diseases, and rare diseases
- Advance next-generation LAT1 inhibitors (Best-in-Class) into clinical trials
- Expand pipeline through the creation of novel transporter inhibitors
- Build a sustainable drug discovery and business platform by integrating diverse drug discovery technologies

| Company name | J-Pharma Co., Ltd |
| Head office |
8th Floor, VORT Hamamatsucho II 1-10-11 Hamamatsucho, Minato-ku, Tokyo 105-0013 TEL: General inquiries (main number) +81-3-6432-4270 Inquiries from investors (Contact IR) +81-3-6450-1146 FAX: +81-3-6432-4271 |
| Founded | December 26, 2005 |
| Representative | Masuhiro Yoshitake (President & CEO) |
| Capital stock | JPY 2,033 million (as of March 31, 2026) |
| Accounting period | March 31 |
| Business description | Research & Development of pharmaceutical drugs |
| Number of employees | 18 (as of March 31, 2026) |

- 40 years of industry experience
- Over 30 years in global leadership roles at major pharma; achieved FDA and EMA approval for 3 drugs
- Successfully completed domestic Phase II study of the company’s lead compound; global Phase III to initiate (FDA agreement obtained)

- 20 years of industry experience
- Global marketing experience at consulting firms and major pharmaceutical company
- Founded a startup, increased its valuation 200-fold, and sold it to a fund
- Led cumulative fundraising of JPY 9B for the company

- Served in various leadership roles at Shimadzu Corporation, including General Manager of the Corporate Strategy Office and President of its European subsidiary
- Appointed Director of Shimadzu Corporation in 2013, while also serving as Executive Officer responsible for sales, finance and treasury, risk management, and Chief Financial Officer (CFO) functions
- Has served as Executive Advisor to Shimadzu Corporation since April 2023
- Appointed Outside Director of the Company in June 2025

- Over 30 years of experience in the biotechnology and pharmaceutical industry, including drug discovery research, antibody therapeutics development, and new business creation at the U.S. National Cancer Institute and Takeda Pharmaceutical Company
- Led hands-on investments in biotech startups at an investment firm/venture capital fund; as Representative of TM Partners, supports commercialization of academic technologies, venture development, and talent development, and has served as an outside director at multiple companies
- Has served as an Outside Director of the Company since 2020 and as an Outside Director (Audit and Supervisory Committee Member) since 2023

- Established Tajima Certified Public Accounting Firm in 2010 and has provided management control and fundraising support primarily to biotechnology startups
- Has served as an Outside Director and Auditor for multiple biotechnology and drug discovery companies, with deep expertise in finance, accounting, and intellectual property
- Appointed Outside Director (Audit and Supervisory Committee Member) of the Company in 2023

- Currently provides broad legal support in corporate law, compliance, and related areas at Kitamura, Kato & Sano Law Office
- Contributed to governance and risk management as Outside Director/Auditor at listed companies
- Joined the Company as Outside Corporate Auditor in 2023 and appointed Outside Director (Audit and Supervisory Committee Member) in October 2023
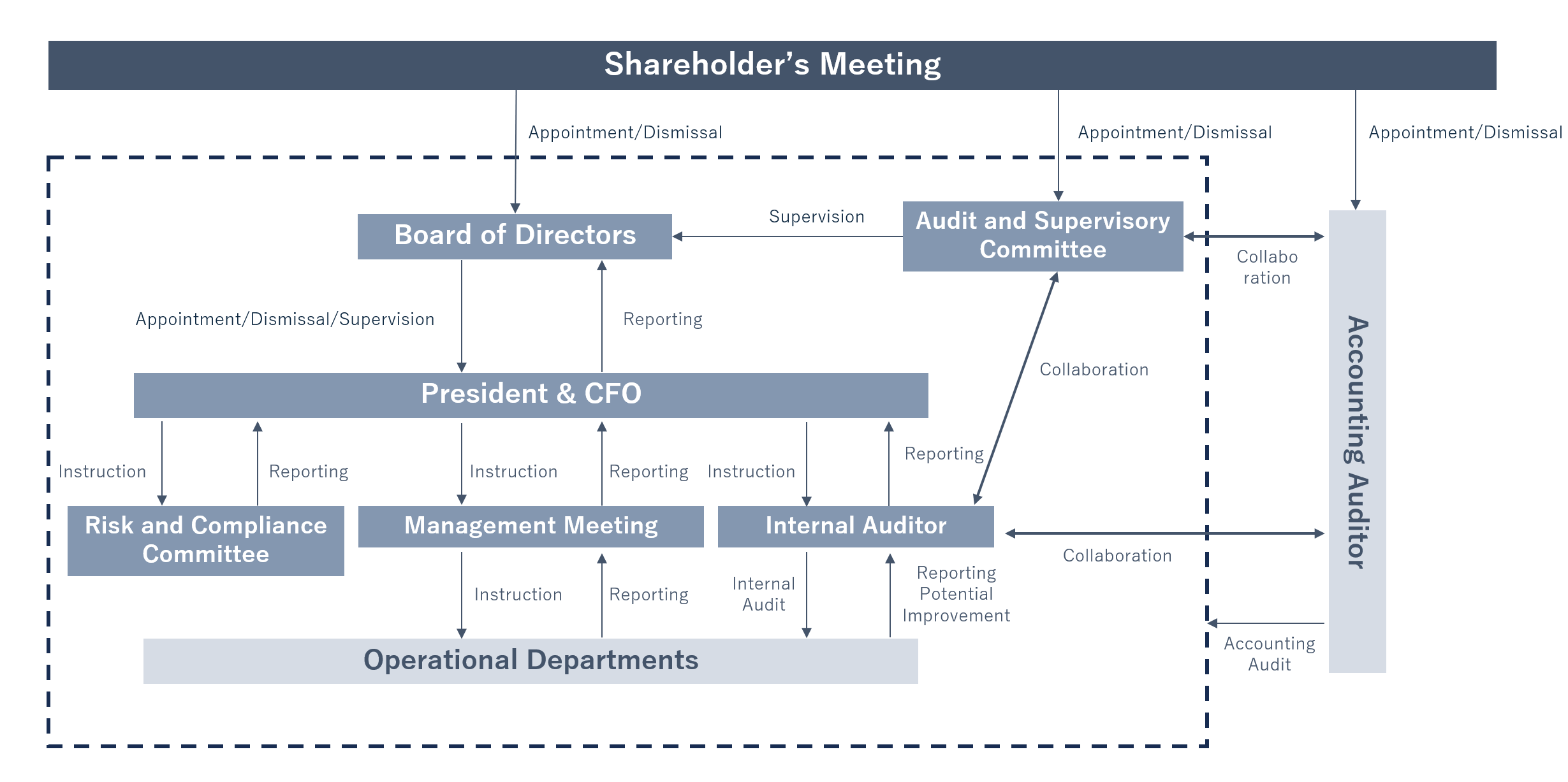
The Company's basic management policy is to ensure management transparency to shareholders and other stakeholders and to continuously increase corporate value by conducting rational and efficient management activities.
The outline of the Company's corporate governance system is as follows.
| Dec 2005 | J Pharma Co., Ltd. was established in Toranomon 1 chome, Minato ku, Tokyo, as a company specializing in drug development targeting membrane transporters |
| Oct 2006 | Awarded the Medical Device Practical Research Support Project funded by the National Institute of Biomedical Innovation for “Development of drug for treating hyperphosphatemia” |
| Oct 2006 | Relocated the head office to Shinjuku 2-chome, Shinjuku-ku, Tokyo |
| Sep 2007 | Awarded Innovation Practical Application Project from New Energy and Industrial Technology Development Organization (NEDO) for “Development of scirrhous stomach cancer therapy” |
| Oct 2007 | Awarded Molecular Imaging Research Program from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) for “Development of novel molecular probes targeting cancer cell-specific membrane proteins” by J-Pharma |
| Aug 2009 | Awarded Innovation Promotion Project from NEDO for “R&D project and clinical phase I study of a novel phosphoric acid adsorbent JPH101” |
| Aug 2010 | Awarded Technology Development for Promoting the Transfer of Basic Research to Clinical Research from NEDO for “Research and Development of a New Anticancer Therapy Targeting Essential Amino Acid Transporter (LAT1) Expressed in Cancer Cells” |
| Apr 2013 | Awarded Venture Practical Application Grant Project from NEDO for “Clinical development of an innovative anticancer drug by inhibiting amino acid transporter” |
| May 2013 | Relocated the head office to Tsurumi-ku, Yokohama, Kanagawa Prefecture |
| Jul 2013 | Awarded a Special Zone Leading Project Grant from Yokohama City for “Development of an in vitro diagnostic kit for triple-negative malignant breast tumor” by J-Pharma |
| May 2014 | Awarded Venture Practical Application Grant Project from NEDO for “Establishing POC of a novel anti-cancer drug JPH203 by clinical trials” by J-Pharma |
| Jul 2014 | Awarded a Special Zone Leading Project Grant from Yokohama City for “Commercial application of in vitro diagnostics for triple-negative breast cancer” by J-Pharma |
| Jan 2015 | Initiated a Phase I clinical trial of the LAT1 inhibitor Nanvuranlat (development code: JPH203) in Japan |
| Jul 2017 | Completed Phase I study of nanvuranlat (Development code: JPH203) |
| Nov 2018 | Started Phase II study of nanvuranlat (Development code: JPH203) in Japan |
| Apr 2019 | Concluded an agreement with Ohara Pharmaceutical Co., Ltd. for licensing and co-development of nanvuranlat |
| Apr 2022 | Obtained Orphan Drug Designation from the U.S. FDA for nanvuranlat (Development code: JPH203) for the treatment of advanced biliary tract cancer |
| Dec 2022 | Completed Phase II study of nanvuranlat (Development code: JPH203) in Japan |
| Oct 2023 | Awarded Fast Forward Commercial Research Grant from the National Multiple Sclerosis Society (NMSS) for the development of the CNS-penetrant LAT1 inhibitor JPH034 for multiple sclerosis |
| Oct 2023 | Established J-Pharma USA for the purpose of talent recruitment to support pharmaceutical development activities in the United States. (liquidated in July 2025) |
| Jun 2024 | JPH034 was selected for the Drug Discovery Venture Ecosystem Enhancement Program of the Japan Agency for Medical Research and Development (AMED) |
| Sep 2024 | Obtained U.S. FDA clearance of the IND application to initiate clinical trials in cancer patients |
| May 2025 | Received positive feedback from the U.S. FDA regarding the CMC (Chemistry, Manufacturing and Controls) of nanvuranlat, confirming that it meets the quality standards required by the FDA at a commercial manufacturing scale |
| Jun 2025 | Relocated the head office to Hamamatsucho, Minato-ku, Tokyo |
| Dec 2025 | Started a global Phase III study of nanvuranrlat (Development code: JPH203) for second-line treatment of biliary tract cancer. |
| Feb 2026 | The IND safety review for JPH034 has been completed in preparation for the initiation of a Phase I clinical trial in the U.S. |





(Japan)

Health Security
(Japan)

School of Medicine
(Japan)

(Japan)

Center
(Japan)
