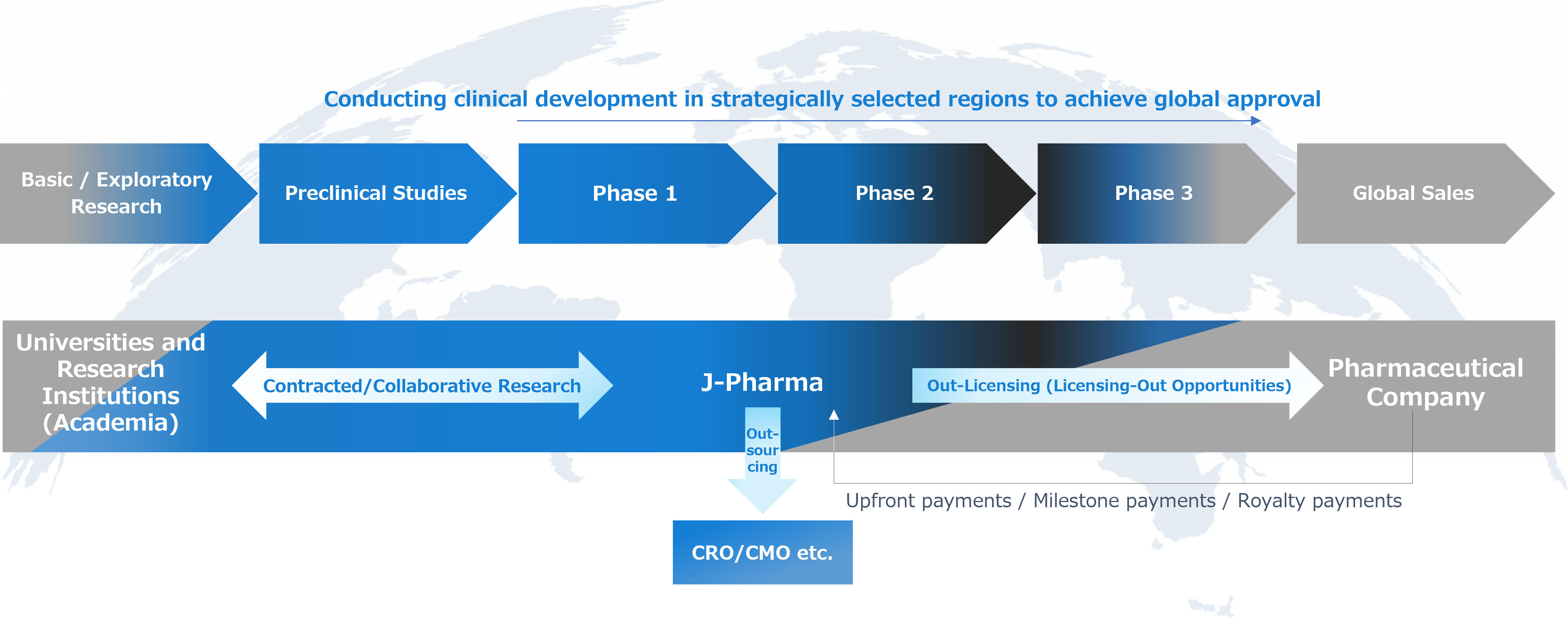
As a global venture company targeting the worldwide pharmaceutical market, we are advancing the research and development of innovative medicines. Our business consists of a single segment focused on drug discovery and development, and our clinical development activities are conducted at appropriate locations and timing, with the objective of obtaining approvals from major regulatory authorities, including the U.S. FDA.
To ensure the successful execution of global development, we have established an organization centered on a management team and R&D professionals with extensive international experience. In addition to working with CROs and consultants in Europe and the United States, we collaborate with leading academic institutions at the forefront of science. Our principal contract research and collaboration partners include Georgetown University in the United States (multiple sclerosis), and the Turku PET Centre (multiple sclerosis). Furthermore, we receive ongoing guidance from globally active experts to enhance our development strategies and research activities, with the aim of delivering results at a global standard.
As a tangible outcome of this organizational framework, our lead compound, nanvuranlat, received Orphan Drug Designation from the U.S. FDA in April 2022. In September 2024, we received IND approval from the U.S. FDA, and in December 2025, we initiated a global Phase 3 trial. Advancing to a global Phase III clinical trial following FDA review based primarily on clinical data generated in Japan is rare, and there are only a very limited number of comparable cases among Japanese biotech ventures.
In addition, for our second compound, JPH034, we received clearance of the Investigational New Drug (IND) application from the U.S. FDA and initiated dosing in the first subject in a Phase I clinical trial in the United States in March 2026. These developments demonstrate our strong development capabilities and global execution strength, and provide a solid foundation for future value creation.
